 
Liquid phase reaction: Except at high pressures Ideal solution Law of mass actionĮquilibrium conversions for single reactions 
Standard-state pressure Po of 1 bar An ideal solution: An ideal gas: f (composition) f (P) f (T).Readily calculated at any temperature from the standard heat of reaction and the standard Gibbs energy change of reaction at a reference temperatureĬalculation the equilibrium constant for the vapor-phase hydration of ethylene at 145 and at 320 ☌ from data given in App.c From Table C.4 The heat capacity data: The equilibrium constant for the reaction, f(T) The standard Gibbs energy change of reaction, f(T) Other standard property changes of reaction :įor a chemical species in its standard state: summation The fugacity of a species in solution: For pure species i in its standard state: Standard Gibbs energy change and the equilibrium constant if not in chemical equilibrium, any reaction leads to a decrease in the total Gibbs energy of the system.the total Gibbs energy at constant T and P is determined by ε.ε is the single variable that characterizes the progress of the reaction.Įquilibrium criteria to chemical reactions the change of the moles of a species ni: integration total stoichiometric number: summationĬonsider a system in which the following reactions occur, if there are present initially 2 mol CH4 and 3 mol H2O, determine expressions for the yi as functions of ε1 and ε2.νi,j : the stoichiometric number of species i in reaction j.Two or more independent reactions proceed simultaneously.If decomposition occurs according to the reaction, find expressions which relate the number of moles and the mole fraction of each chemical species to the reaction coordinate ε. Determine expressions for the mole fractions yi as functions of ε.Ĭonsider a vessel which initially contains only n0mol of water vapor. A differential change in the number of moles of a reacting species: Reaction coordinate, which characterizes the extent or degree to which a reaction has taken place.įor a system in which the following reaction occurs, assume there are present initially 2 mol CH4, 1 mol H2O, 1 mol CO and 4 mol H2.The purpose of this lecture is to determine the effect of temperature, pressure, and initial composition on the equilibrium conversions of chemical reactions.Although reaction rates are not susceptible to thermodynamic treatment, equilibrium conversions are. 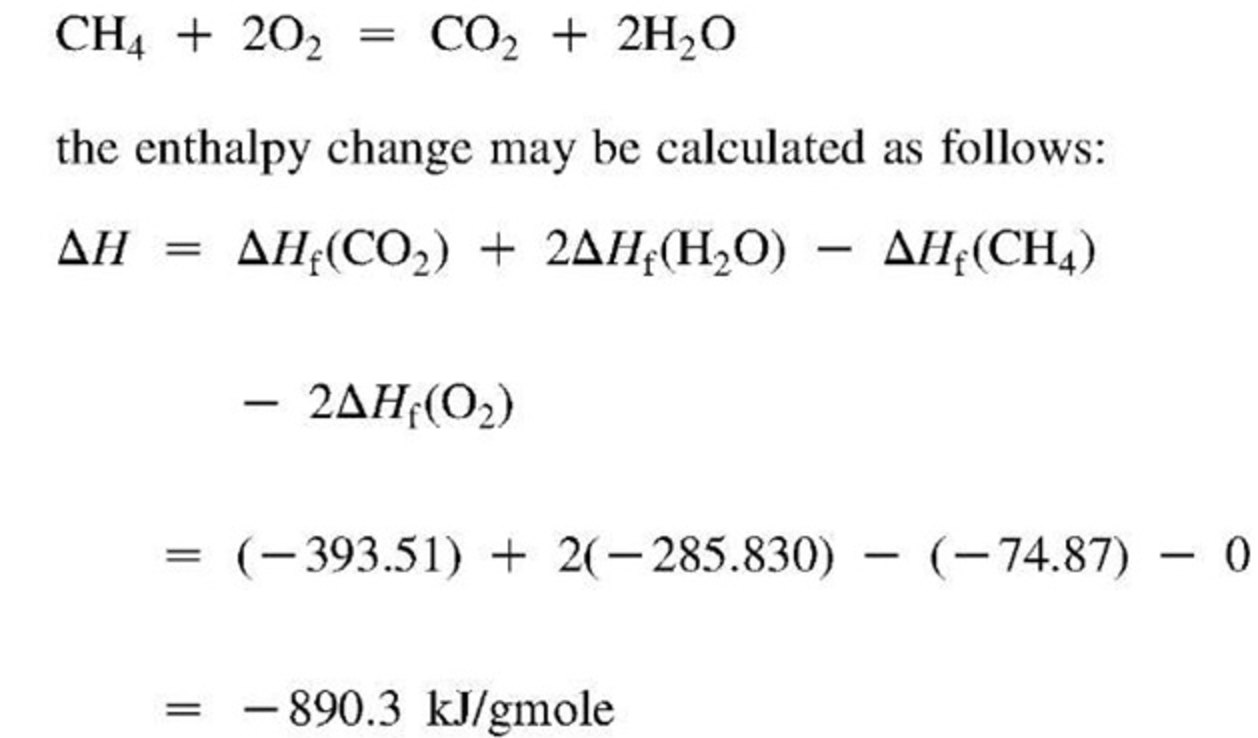
Both the rate and equilibrium conversion of a chemical reaction depend on the temperature, pressure, and composition of reactants.Advanced ThermodynamicsNote 12Chemical-Reaction Equilibria Lecturer: 郭修伯 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
